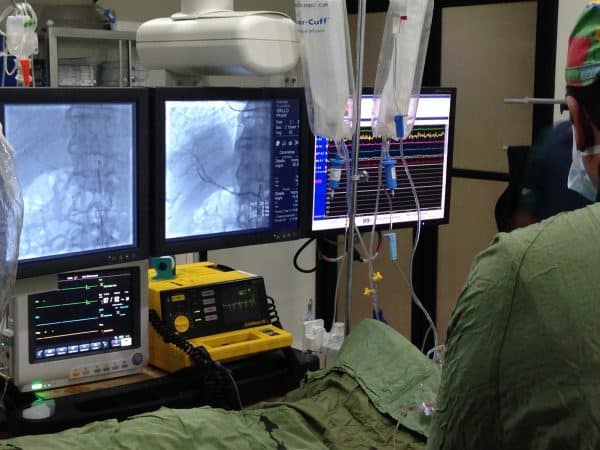
Facing death inspires desperate measures. Like having your stem cells drawn in Florida, sent to Israel for processing, and shot into your ailing heart in the Dominican Republic. How far would you go to save your own life?
By Mark Cohen, Photography By Mauricio AlejoStem Cell Therapy by Doctor Zannos Grekos MD – Among leading stem cell doctors in the USA. Ron O’Leary, 36, has never felt so exposed. Naked from the waist down, the 6’2″, 210-pound man is lying atop an x-ray table, shivering in the air-conditioned chill. His blue eyes are fixed on the flexible 4-foot catheter about to be threaded through his femoral artery to his heart. He’s flown 1,000 miles from his home in Florida to the Dominican Republic because parts of his heart are dead.Now, an ECG broadcasts the plodding clunk-clunk of his scarred heart, which is pumping at 30 percent of its normal capacity. The nurses begin slathering his privates with a dark-brown antiseptic solution. Thoughts flit through his mind. Boy, is that cold . . .
Is this going to hurt? . . .
I just want my heart back . . .
It’s 2 p.m. in the dimly lit fourth-floor cardiovascular department at Centro de Otorrino, a private hospital in downtown Santo Domingo. O’Leary suffers from idiopathic dilated cardiomyopathy, a condition in which the heart muscle has stretched and then becomes weak and starts dying off. His heart is so damaged that it struggles to pump blood to his lungs, leaving the former restaurant owner unable to work and barely able to walk 40 yards without wheezing. His doctors in Florida counseled him that he needed to adjust to this new reality and prepare himself for a heart transplant in a few years if his condition deteriorated. Faced with this dramatic decline in his quality of life, O’Leary, who had been in good health 11 months prior, has flown to one of the poorest countries in the Caribbean for a new type of cardiac therapy. Doctors are going to use O’Leary’s own stem cells to repair his heart. There’s only un problemita. It’s not legal, at least in the United States, because the U.S. Food and Drug Administration classifies the adult stem cell as a high-risk biologic product, and requires a rigorous review of its safety and effectiveness before it can be marketed. Prior to leaving home, O’Leary had signed 12 pages of waivers exempting the doctors who would be inserting the catheter from any kind of malpractice suit.
As a nurse places a sheet over O’Leary’s groin, Dominican cardiologist Roberto Fernandez de Castro, M.D., picks up a polypropylene syringe from a stainless-steel surgical tray and gives it a slight shake. Inside, suspended in a yellow slurry of plasma, are 95 million of O’Leary’s stem cells. They were grown from his own blood, which was extracted a week earlier at the offices of Regenocyte, the Naples, Florida-based company overseeing this procedure.
Next, Dr. Fernandez screws this small syringe into the opening of a foot-long black plastic syringe. It’s then attached to the specially designed catheter that his partner, Hector Rosario, M.D., has begun feeding through a spot on O’Leary’s upper-right thigh, up into the lower-left chamber of his heart. On the black-and-white fluoroscopy monitor, the catheter snakes into view. Its tip flicking in and out in response to Dr. Rosario’s touch, the needle probes for a fertile site on the interior wall of O’Leary’s left ventricle. Dr. Rosario turns a dial at the end of the syringe to lengthen the tip, and with the help of a plunger, implants the stem cells directly into O’Leary’s heart.
“Ouch . . . pain, definitely a little pain there,” says O’Leary.
On the ECG, O’Leary’s rhythm spikes into a pattern of rapid squiggles. Dr. Fernandez mutters under his breath in Spanish, and Dr. Rosario adjusts his grip on the needle. “That’s actually a good sign,” says Dr. Rosario. In his broken English, he explains that the pain shows there’s enough healthy tissue in the heart muscle to react to being jabbed, which should help the stem cells take root and start growing new blood vessels. Then he maneuvers the needle to a second spot on the ventricle wall and fires off another 3 million cells.
Despite all the billion-dollar research, medical advances, and new life-extending recommendations of the past 50 years, there still isn’t much you can do for a damaged heart. If you suffer a severe heart attack or an enlarged heart like O’Leary’s, your choices are limited: Downshift your daily life so that watering the garden ranks as your most strenuous activity, or get in line with the other 3,000 Americans waiting for one of the 2,200 donor hearts available annually. The human heart may be one of our most mechanically sophisticated organs, but it’s just not a good healer. With its own electrical system and unique muscle tissue beating 100,000 times a day, the heart is just too complex and too busy to repair itself significantly. Deprive critical cardiac tissue of oxygen, even for just a few minutes, and it dies. Weaken a single chamber or a single valve, and pumping efficiency plummets. Allow the damaged parts to dip below a certain baseline level of functioning, and they only become worse, never better.
Which is why stem-cell therapy is so intriguing—and potentially so lucrative. Of the 17 million adults in the United States who have suffered heart attacks or other major cardiac malfunctions, 5.7 million have started on the inexorable slide toward heart failure. And more than 3 million of them are men. If science could harness the regenerative powers of the patients’ own stem cells to reverse that slide, the savings in terms of lives and dollars would be enormous. It’s nothing short of the groundbreaking first step on the path toward growing your own replacement heart. “Stem-cell therapy shifts the whole paradigm for treating heart disease, whether it’s caused by heart attack, arrhythmia, or cardiomyopathy,” says Joshua Hare, M.D., a cardiologist and the director of the University of Miami’s interdisciplinary stem-cell institute. “My prediction is that in 10 to 15 years, thanks to this new ability to repair damage, heart disease will no longer be the number one killer in the country.”
Dr. Hare is overseeing four of the 21 phase 1 and phase 2 cardiac stem-cell-therapy clinical trials now under way at 43 research institutions around the country. While scientists race to win approval from the FDA, they’re playing by the rules—they’re following the systematic, government-regulated route of testing experimental medical treatments to make sure they’re safe before they can become standard practice. Researchers estimate the cost of a three-phase stem-cell trial will be upward of $50 million, with a 10-year time table. Regenocyte and the nearly two dozen other for-profit off shore stem-cell clinics are taking a shortcut, but at whose expense?
Since Regenocyte began offering treatments in the Dominican Republic in February 2008, its doctors have performed 75 of what it calls myocardial cellular regeneration procedures, at a cost of $64,500 a pop — not a penny of which is covered by insurance. The process kicks off in Naples, Florida, with a blood draw; the blood is then immediately whisked by courier to the lab of Regenocyte’s affiliate, TheraVitae, in Tel Aviv, Israel, for centrifuging and culturing. After a 5-day incubation period, which increases the stem cells’ numbers a thousandfold, a TheraVitae representative boards a flight with the stem cells in a temperature-controlled tissue container, and arrives in Santo Domingo on the morning of the procedure. Shuttling cells back and forth across six time zones and performing the procedure outside the United States allows the company to operate without interference from U.S. authorities. The FDA’s jurisdiction extends only to products manufactured within the United States; as it is, the only step of the process the company carries out on American soil is a needle stick to the arm.
“It’s a questionable practice,” says Emerson Perin, M.D., Ph.D., the director of clinical research for cardiovascular medicine at the Texas Heart Institute, who’s supervising five of the clinical trials. “They’re charging big bucks for an experimental procedure that will require years of rigorous research before we know if it will benefit patients. It’s a total gunslinger mentality, and that’s coming from someone in Texas.” Concerned about the rise in “stem-cell tourism,” the nonprofit International Society for Stem Cell Research recently cardiac-produced a handbook for patients who are considering experimental therapies. It outlines the risks, which are physical, financial, and psychological.
The greatest danger in adult stem-cell cardiac treatments derives from the fragility of patients with terminal heart disease. “They can die if they sneeze the wrong way,” says Amit Patel, M.D., the director of clinical regenerative medicine in cardiac surgery at the University of Utah, who is overseeing a phase 2 clinical trial using stem cells to treat dilated cardiomyopathy. If a patient’s weak condition causes some other medical problem, many smaller clinics simply don’t have the resources to provide emergency help, he says. A further concern for patients is the cost of that emergency care and the burden of additional fees.
That said, the technique and cells Regenocyte’s doctors are using are low risk. “This modified catheterization approach has had few complications,” says Dr. Patel. However, as with any heart catheterization, there is roughly a 1 percent chance of perforation of the heart, which would likely result in death, he says. Dr. Patel also notes that although adult stem cells have been used in phase 1 and phase 2 clinical trials, long-term concerns about safety and efficacy remain, because there is no long-term data yet.
Even when patients understand and accept these risks, they still need to ask the next question: Does the treatment actually work as advertised? A peer-reviewed clinical trial is the gold standard for verifying efficacy. The consensus in the medical establishment is clear, as Clyde Yancy, M.D., president of the American Heart Association and medical director of the Baylor University heart and vascular institute in Dallas, explains: “We do not encourage patients to pursue any therapy that has not met the standards of evidence-based medicine or is not a part of an established study.” If the treatment doesn’t work, patients find themselves both out of money and out of options, which can be psychologically devastating.
There is also a concern that what companies like Regenocyte are doing creates a dangerous precedent that could lead to patients rolling the dice with other stem-cell therapies that have less scientific merit. “There’s no way you can let stem cells go unregulated,” says Doris Taylor, Ph.D., the director of the cardiovascular repair center at the University of Minnesota stem-cell institute. And yet, perhaps because Taylor is so familiar with stem cells’ potential, she also makes an interesting concession for a scientist at an elite research institution. Asked if she might advise someone she loved who was dying of heart failure to book a flight to the Dominican Republic, she replies, “You know, I’m not sure. But I would discuss it first with my colleagues.”
Adult stem cells usually aren’t associated with controversy. Embryonic stem cells, the kind grown from cryo-frozen fertilized eggs stored in liquid nitrogen in fertility clinics, are the ones that have been at the flash point of moral agonizing. The use of these cells to find cures for Parkinson’s and Alzheimer’s has been hotly debated in presidential elections, taken up by celebrity advocates like Michael J. Fox, and recently restarted by President Obama with the approval of the first cell lines available for research using federal funding. But something curious happened while the ethics debates raged: The cardiac research community’s enthusiasm for embryonic stem cells waned. “Many of us found the whole embryonic-cell debate to be overstated,” says Dr. Hare. “The cells are unpredictable and have the tendency to turn cancerous. They’re almost too powerful.”
So doctors began shifting their research to a more reliable and readily available type of stem cell. Like days-after-conception stem cells, adult stem cells can morph into virtually any other kind of cell, but they’re found in the blood, bone marrow, and many organs of every living human. “Ten years ago, we didn’t even think organs like the heart had stem cells or were capable of regeneration,” says Dr. Hare. “We now realize that the body has a much greater capacity for rejuvenation. It just needs some help.”
Dr. Hare’s office is on the eighth floor of a glass tower near downtown Miami. From there it’s a 3-hour drive west through the Everglades to Regenocyte’s Naples headquarters. On a monthly basis, Dr. Hare finds himself competing for prospective trial participants who have heard the Regenocyte pitch on organ-transplant blogs or on the radio ads the company airs. “What I find frustrating is that they’re preying on people’s desperation. Stem cell therapy has great potential, but there’s so much about it we’re still figuring out.”
For the past 5 years, Dr. Hare and his colleagues have been struggling to answer a host of important questions: What’s the correct dosing, the optimal delivery method, the ideal wait time between heart attack and treatment? Another pressing area of exploration: Which of the hundreds of types of stem cells will prove the most practical and effective for cardiac repair? Dr. Hare and Dr. Patel have been focusing on one especially promising type of bone-marrow-derived stem cell. Another group at the University of Pittsburgh is investigating the stem cells found in fat tissue, which may lead them down the mind-bendingly circular road of one day curing patients’ heart conditions with a by-product of their own liposuction procedures.
Exactly how stem cells work their biological magic is still not fully understood. “We think the cells might send out signals to other cells to migrate to the area to help with the repair,” says Dr. Hare. “Or they might awaken the stem cells in the heart itself.” Another thing researchers need to determine before the FDA can approve stem cell therapy for general consumption is how well they work.
A landmark study published in the December 2009 Journal of the American College of Cardiology showed definitively for the first time that treating a patient with adult stem cells after a heart attack can repair—not just manage—damaged heart tissue. The double-blind, placebo-controlled study, part of a phase 1 clinical trial led by Dr. Hare, found that the patients had significant improvement in heart and lung function and experienced lower rates of side effects, such as cardiac arrhythmia. For instance, the patients averaged a 6.7 percent gain in their heart’s pumping function. Called “ejection fraction,” this common measure of heart function records the percentage of blood flowing into the left ventricle that’s pumped out with each heartbeat. A healthy heart should expel 55 percent to 70 percent of the blood that flows into it. The heart-attack victims in the study averaged an ejection fraction of 50.4 prior to treatment. So a 6.7-point bump puts them back into the normal category.
Another recent study, a multicenter phase 2 trial of 167 heart-attack patients presented at the meeting of the American Heart Association, found that those treated with stem cells added 120 seconds to the length of time they could walk briskly on a treadmill. Each 60 seconds of treadmill improvement is thought to equal about 15 minutes of normal activity, says lead researcher Douglas Losordo, M.D., a professor of cardiology at Northwestern University. That 120 seconds was just an average (one outlier saw a nearly 10-minute gain), but is not insignificant when it extends your range from the distance between bed and bathroom to a stroll around the block.
The typical adult-stem-cell trial is divided into three equal groups: patients who receive the optimal dose, those who receive a reduced dose, and the placebo group. That means if you’re a study participant, the chances of your damaged heart receiving a cutting-edge application of sugar water is 33 percent. It usually takes 12 to 18 months to even find out which group you’re in. “Most people are okay with that,” says Dr. Hare. “They understand that when you walk into a pharmacy and put an FDA-approved pill in your mouth that you’re 99 percent sure is safe and effective, it’s because generations of people before you went along with this approach. But I do hear some who say, ‘I understand, doc, but I don’t want to take my chances. I just want the cells.’ ”
Like many of the estimated 300,000 American men under 55 suffering from some kind of cardiomyopathy, Ron O’Leary had next to no warning of the illness that would turn his high-functioning heart into a swollen lump of scar tissue. With his outgoing personality, mop of blond hair, and nose bent from an old football injury, he reminded people of a stocky Owen Wilson. Suddenly, in September 2007, he noticed his energy level dropping and had difficulty catching his breath during his long shifts at the restaurant. After several days, O’Leary went to a hospital emergency room, was told he had pneumonia, and left the ER clutching a prescription for an antibiotic. A week later he was coughing up blood and went to an ER at a second hospital. Doctors there ran an ECG and admitted him for additional tests.
The diagnosis: idiopathic dilated cardiomyopathy. The doctors couldn’t even be sure what had caused O’Leary’s heart condition, because it didn’t run in his family. The most likely culprit was a viral infection. One doctor told O’Leary his was the weakest heart he’d ever seen. It was emptying just 8 percent of the blood that flowed into it before his doctors loaded him up with diuretics, beta-blockers, and ACE inhibitors, which boosted his ejection fraction to 30 percent.
“Numb. Just floored,” O’Leary recalls of his reaction. “I mean, I’d never even really been sick before. The only medical professional I saw on a regular basis was my dental hygienist. My cardiologist put in a defibrillator, kept me on the medication, ordered regular tests, and told me I might eventually need a transplant.”
And under most circumstances, that would have been that. But O’Leary lives in Sarasota, close to the burgeoning South Florida stem-cell belt. After an article about his plight appeared in a local newspaper, O’Leary received an unsolicited call from a satisfied Regenocyte customer. Two months later he attended his first informational seminar and was, as he describes it, just blown away by the patient testimonials. (See “5 Stay-Safe Rules for the Desperate Patient.”)
Regenocyte’s founder, 44-year-old Zannos Grekos, M.D., is an experienced interventional cardiologist who estimates he has performed more than 10,000 angioplasties, angiograms, and pacemaker implants. Early in his career, Dr. Grekos and the hospital he worked in were sued for malpractice after one of his patients died. The woman’s family claimed he waited too long to refer her for a coronary bypass after it should have been apparent that a cardiac catheterization he’d performed hadn’t worked. Dr. Grekos’s insurance company settled the case out of court for $600,000 in 2001. It’s not uncommon for interventional cardiologists to face such suits, because these procedures are high risk. But the incident left the young physician cynical about the legal and regulatory aspects of his business. “It can make you question whether you want to be in a profession where you do everything you can to help a patient, work 100 hours a week, and end up having lawyers still question what you do,” he says.
Four years ago, Dr. Grekos was working at his cardiology practice in Naples, Florida, when a longtime patient, a prominent local businessman, phoned to tell him that he’d scheduled to have stem cells injected into his heart. In Bangkok. “I said, ‘You scheduled what? ‘ ” says Dr. Grekos. But he checked into the company—TheraVitae, which had state-of-the- art facilities in Bangkok and Israel—and decided it looked legit. “So I told him, ‘I’ll go over with you, but if anything doesn’t look quite right, you’re taking the next plane home.’ Then I met the doctors and realized it was the most fantastic development I’d ever seen. I also realized that there were things about it we could do better if we licensed the technology ourselves and performed the catheterizations closer to home, saving patients from sitting on a plane for 36 hours.”
Dr. Grekos studied pioneering research being done by Dr. Patel (then at the University of Pittsburgh), Dr. Perin at the Texas Heart Institute, and Dr. Losordo (then at Tufts), among others. Various pilot studies examining the safety and efficacy of injecting adult stem cells into the heart, either by surgery or through a modified angioplasty catheter, indicated that stem-cell treatments held promise. Dr. Grekos believed using a catheter that allowed him to implant the cells directly into the heart wall might work better. He found a Florida-based company called Bioheart that had created the MyoCath, a catheter with a retractable needle tip to implant stem cells, and trained to use it at the University of Florida in 2008. (A phase 2 clinical trial treating patients with heart failure using the MyoCath and delivering stem cells derived from thigh muscle is under way at the Duke University heart center.) Based on what he learned in Thailand and from other research, Dr. Grekos decided to use the MyoCath with stem cells cultured by TheraVitae. He teamed with Dominican cardiologists and trained them to use the MyoCath.
How does Dr. Grekos respond to those who say he is exploiting desperate patients and who question his ethics for charging for an experimental treatment? “I’m just trying to help my patients,” says Dr. Grekos. “In our experience, we see a 21-point ejection fraction gain. I actually think it would be more unethical to administer a placebo.” However, until Dr. Grekos publishes the data in a peer-review forum, there is no way for anyone to really know if these numbers are accurate.
In the one pilot study that Dr. Grekos has made public, he presented data about 20 ischemic cardiomyopathy patients at the New Cardiovascular Horizons conference in 2008. The group’s baseline average ejection fraction was 28 percent (range 14 percent to 42 percent) at the start. After treatment and 6 months, it was 49 percent (range 38 percent to 56 percent). He says he’s compiling more data and that he’s waiting until he has the results from 100 patients, including follow-up stats after a year, before publishing it. But on average, he says, he’s still seeing a 21-point gain.
Some cardiologists, such as Dr. Patel, who is conducting a stem-cell cardiomyopathy trial, believes the treatment could be that successful. “Most heart-attack patients have very little change in their ejection fraction, but these heart-failure patients have 10 percent to 30 percent,” says Dr. Patel. “When you start with patients with more-advanced heart failure, such as cardiomyopathy, there’s a lot more room for improvement. Regenocyte could be seeing gains that high.”
To the researchers who say Regenocyte’s $64,500 fee is double what the procedure should cost, Dr. Grekos counters, “How do they know what our expenses are? The cell culturing and couriering back and forth to Israel alone costs $20,000. I’m just glad we’re making some profit on it, because if not, the treatment wouldn’t be available.” He also notes that Regenocyte has an impeccable safety record and that no patients have died during the procedures.
Dr. Grekos even tries to turn the tables on the FDA by invoking “the practice of medicine,” the long-held precedent granting physicians wide latitude in applying established procedures in new combinations. He says his treatments are little different from those of a physician who orders a special formulation from a compounding pharmacy or prescribes the off-label use of a drug approved for one condition to treat another. “It’s the same way in vitro fertilization developed in this country. It was never ‘approved’ by the FDA. It developed as the field established its own standards and governed itself under the practice of medicine. I don’t see how adult stem cells, which are a patient’s own cells and not even mixed with someone else’s, should be treated any differently than IVF was treated.”
This sounds reasonable, but from the FDA’s perspective, it flies in the face of logic. “Cellular therapies that are more than minimally manipulated, even if they’re used in the same person they come from, are regulated under the Public Health Services Act and require premarket approval,” says FDA spokeswoman Karen Riley. As soon as you start taking cells from one part of the body, adding things to them to make them grow, and putting them in the heart, you’re doing more than minimal manipulation in the eyes of the FDA. Doctors are in essence functioning as drug manufacturers when they prepare and use these stem-cell treatments, so they are subject to federal regulations.
The FDA is making the same argument against Regenerative Sciences, a for-profit stem-cell company (in the relatively low-risk arena of joint and disc repair) that treats its patients within U.S. borders. When in July 2008 the FDA informed the Denver-based clinic that it couldn’t promote its stem-cell treatment like a drug treatment, Regenerative responded by filing a suit saying that what it was doing was out of the agency’s jurisdiction. The case is now in early pretrial proceedings in U.S. District Court in Colorado.
Dr. Grekos says he has already begun planning for the day when stem-cell therapy becomes available in the United States, and he is working on a two-pronged strategy: moving treatment for any approved uses to his cardiology practice in Naples, and continuing to use unapproved treatments off shore. He says he just gained approval to treat patients in Freeport, in the Bahamas.
Recognizing that little can be done to stop the off shore clinics, and in an effort to review those clinics’ claims, a group of scientists and doctors recently teamed up to create the International Cellular Medicine Society (ICMS). At this online registry, off shore clinics have patient results tracked and verified by an objective third-party source. But of 20 off-shore clinics contacted for a survey, only 11 responded, and only one met the criteria used by the ICMS to ascertain safety: TheraVitae. The report concluded that the most troubling aspect about the stem-cell clinics was a lack of transparency: “Nearly all the evaluated clinics refused to answer all questions posed.”
Regenocyte did not participate because the survey was only for companies that process their own stem cells. But Regenocyte is not sharing its results with the ICMS. “When I first talked to Dr. Grekos last summer, he seemed interested,” says Christopher Centeno, M.D., the Denver pain-management specialist and ICMS member who founded Regenerative Sciences. “Then he had me talk to his business manager, who later got back to me and said they’d decided not to participate. The way he put it to me was, he just didn’t see what’s in it for them.” By joining such a registry, Regenocyte would be opening up its treatments to the same statistical metrics used by clinical trials. Patients who failed to respond as favorably would be lumped in with the star performers who achieved 10-minute gains in their treadmill times. The focus of prospective patients could start to shift from the anecdotal evidence that now dominates the company’s marketing to the law-of-averages bottom line.
Ron O’Leary answers the phone. He sounds out of breath. “Hey . . . How are you doing?” he says, his voice ragged and halting. “Sorry,” he goes on. “I just got in from a bike ride.” It’s been 6 months since his stem-cell procedure and O’Leary, now 37, sounds to be settling back into his old routine. That is, his old old routine, the one he had before a freak-of-nature heart malady reduced him to puttering around like a geriatric.
A few weeks earlier, O’Leary traveled to Naples for his 6-month follow-up. His ejection fraction had increased to 44, which was 14 points above where it was before the doctors repaired parts of his heart. He didn’t experience the 21-point jump that Dr. Grekos says is typical, but 14 points is a significant improvement.
For O’Leary, though, the most telling test may have been one he administered to himself a week before his office visit. He’d ventured out onto a local bike trail for a 20-mile ride with two friends. “It’s not like I was racing Lance Armstrong; these guys are in their 50s,” he says. “On the way back I took off for a stretch. I left them behind. That’s when I knew. I feel like there’s nothing I can’t do.”
5 stay-safe rules for the desperate patient
If conventional medicine fails, use these guidelines to find an alternative—whether it’s an off shore therapy, a clinical trial, or a holistic approach.
1. Seek a second opinion
Discuss with your primary-care physician any unsanctioned treatment you’re considering. He or she knows the full history of your condition, says Sam Moon, M.D., M.P.H., a Duke integrative-medicine clinician. And unlike the doctor administering an unsanctioned treatment, your doctor can help you weigh the pros and cons.
2. Disregard patient testimonials
“Patients’ stories are not objective,” says Heather Rooke, Ph.D., science director for the International Society for Stem Cell Research. Even when improvement does occur, it may be independent of the treatment or exaggerated, she says. Ask how the clinic measures improvement and if an independent committee has reviewed the treatment.
3. Investigate your treatment
Surf the WHO Registry Network (who.int/trialsearch), or the NIH clinical trials registry (clinicaltrials.gov) for human clinical research on the treatment. For disease-specific therapies, check the trial’s inclusion criteria—participants should share your age, diagnosis, and treatment history, says Jeffrey Karp, Ph.D., a stem-cell researcher at Harvard’s medical school.
4. Check beyond credentials
The title “doctor” is vague, says Zhaoming Chen, M.D., Ph.D., executive board chairman of the American Association of Integrative Medicine. Contact your state medical board—or if abroad, the department of health—to confirm a physician’s credentials, licensure, and specialty. Ask how many times the doctor has performed the procedure: You don’t want to be among first 50 guinea pigs.
5. Be prepared for the worst
No procedure is risk-free. The key: “Balance hope for long-term gains against the risks of treatment,” says Dr. Moon. Make sure your patient consent form clearly outlines the care and potential side effects, and allows you to withdraw from treatment. Check the clinic’s capacity for emergency care and find out what follow-up is provided.—laura roberson
How stem cells can repair your heart
To find out which of the stem-cell therapies being tested are the most promising, we surveyed key researchers. Here are their predictions.
If you suffer dilated cardiomyopathy
Amit Patel, M.D., a researcher at the University of Utah, is overseeing a clinical trial that he expects will enter its final phase in July 2010. Patients have 200 million of their own cultured bone-marrow stem cells injected into their heart muscle through a catheter. The stem cells may secrete growth factors, and then other cells in the heart help out by remodeling scar tissue, growing new blood vessels, and boosting heart-muscle function.
Estimated time to FDA approval: 2 years
If you survive a heart attack
Jay Traverse, M.D., a cardiologist at the Minneapolis Heart Institute, is using bone-marrow-derived stem cells to treat people within 3 to 7 days of a heart attack. In his phase 1 and phase 2 clinical trials, doctors infuse 150 million stem cells into patients’ hearts using a modified catheter. The stem cells may improve the efficiency of their hearts by growing new blood vessels.
Estimated time to FDA approval: 5 years
If you have heart failure
Joshua Hare, M.D., director of the interdisciplinary stem-cell institute at the University of Miami, is leading a phase 2 trial to treat heart- failure patients. The patients have 20 million to 200 million bone-marrow stem cells, either theirs or ones derived from a donor, put into their hearts through a catheter. The benefits are due to a combination of blood-vessel regeneration, new cardiac-tissue growth, and scar-tissue reduction.
Estimated time to FDA approval: 4 to 5 years
If you have heart disease
Douglas Losordo, M.D., director of cardiovascular regenerative medicine at Northwestern University, recently completed a phase 2 trial of patients suffering from chronic chest pain due to blocked arteries. Stem cells har vested from the patients’ blood are injected into their hearts and arteries through a catheter. The stem cells improve bloodflow by stimulating the growth of new blood vessels and expanding the diameter of coronary arteries.
Estimated time to FDA approval: at least 5 years
If you need a heart transplant
Doris Taylor, Ph.D., director of the cardiovascular repair center at the University of Minnesota stem-cell institute, is using human heart stem cells to grow live hearts on cadaver scaffolds. The cadaver heart is stripped of cells and then stem and progenitor cells are introduced. A beating heart develops in about a week. She’s working on animal studies and expects the first human implant trial to start in a few years.
Estimated time to FDA approval: 10 years—heather hurlock




7 thoughts on “My Illegal Heart – Stem Cell Therapy”
cialis 5mg best price
cheap brand name cialis
Качественные WordPress ссылки в комментариях от 5000 уник. доменов заказать здесь .
Quality WordPress links in the comments from 5000 uniques. domains order here.
I don’t need to tell you how important it is to optimize every step in your SEO pipeline. But unfortunately, it’s nearly impossible to cut out time or money when it comes to getting good content. At least that’s what I thought until I came across Article Forge… Built by a team of AI researchers from MIT, Carnegie Mellon, Harvard, Article Forge is an artificial intelligence (AI) powered content writer that uses deep learning models to write entire articles about any topic in less than 60 seconds. Their team trained AI models on millions of articles to teach Article Forge how to draw connections between topics so that each article it writes is relevant, interesting and useful. All their hard work means you just enter a few keywords and Article Forge will write a complete article from scratch making sure every thought flows naturally into the next, resulting in readable, high quality, and unique content. Put simply, this is a secret weapon for anyone who needs content. I get how impossible that sounds so you need to see how Article Forge writes a complete article in less than 60 seconds! order here.
Use artificial intelligence to cut turnaround time, extend your budget, and create more high-quality content that Google and readers will love. How It Works? WordAi is extremely fast and intuitive. Just enter your content, click rewrite, and in a matter of seconds, WordAi will rewrite an entire piece of content for you. WordAi comes up with different ways to express the same ideas by rewriting every sentence from scratch. This not only removes duplicate content, it also makes the rewritten content read completely naturally. Scale Your Business Make your entire content production process 10x more efficient. Use AI to create more high-quality, unique content that impresses clients and engages readers, all without having to put in more hours Register here and get a bonus.
retin-a micro gel